thermodynamics - Confused why delta ∆Q and dQ don't make sense for heat Q - Chemistry Stack Exchange
Por un escritor de hombre misterioso
Descripción
In my chemistry teacher's notes, some notations concerning the heat $Q$ are marked as inappropriate.
$Q$: yes
d$Q$: no
$\delta Q$: yes
$\Delta Q$: no
In the second bullet in the screenshot below

Essentials of Computational Chemistry

Full article: Theory of cross phenomena and their coefficients beyond Onsager theorem

Tackling a Century Mystery: Entropy

Putting It Into Reverse – Watts Up With That?
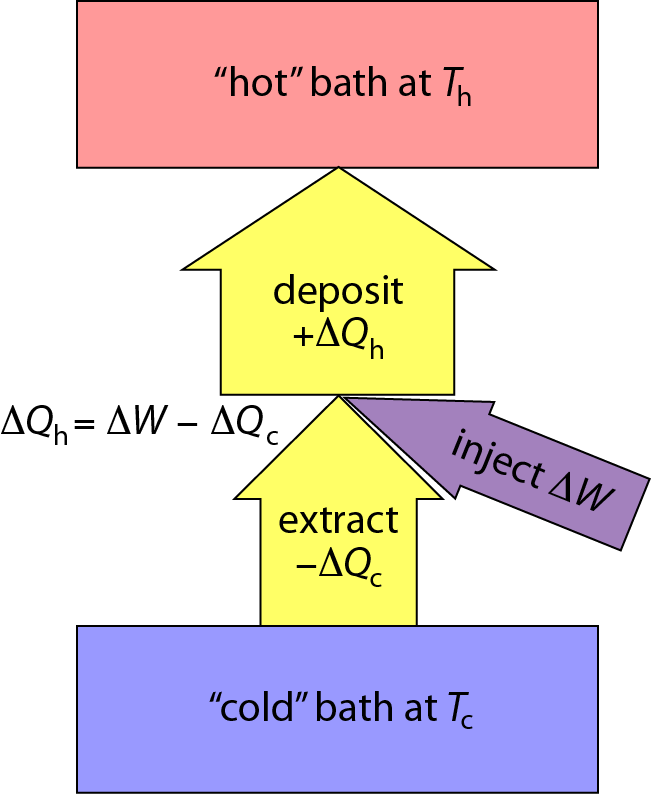
Heat Pumps Work Miracles


In which ways are the central limit theorem and the second law of thermodynamics related? - Quora

Can the Second law of thermodynamics be abandoned?

Thermodynamics Internal Energy (E) of a mineral = Sum of energy stored - ppt download
de
por adulto (el precio varía según el tamaño del grupo)






